Introduction
Around 2,600 courses of chemotherapy are given in Lothian, UK, each year, to people with a range of cancer types, at all stages of disease and with different treatment intent. Decision-making around chemotherapy and other systemic anticancer therapy (SACT) can be challenging, both for clinical teams as well as their patients and those close to them. This is particularly true in the palliative setting, where the cancer cannot be cured but where there still may be significant benefits from oncological therapy. However, the risk of treatment-related morbidity and mortality is real1 and this should be understood by patients, their families and carers.
It is known that patients do not always have a good grasp of the extent of their cancer and the implications of this for their future;2 one such example, often misunderstood, is the fact that chemotherapy given for advanced cancer will not offer cure.3 A further consideration, often also poorly understood, is that even where chemotherapy does extend life, a good quality of life for this additional time is not a given.4 Concerns about the potential for overtreatment in advanced cancer are not new.5 However, with the spotlight on shared decision-making6,7 and in the knowledge that chemotherapy and other SACT can carry significant burden to patients and those close to them, understanding how people feel about the decision-making process is of great interest.
The aim of this study was to explore how patients who had undergone palliative chemotherapy at the Edinburgh Cancer Centre (ECC), UK, felt, with hindsight, about their decision; in particular, to gauge levels of ‘decisional conflict’, defined as ‘the state of uncertainty about the course of action to be taken when choice amongst competing actions involves risk, loss or challenge to personal life values’.8 High levels of decisional conflict are concerning, with uncertainty suggesting that the patient may not have felt confident about the decision they made.
Methods
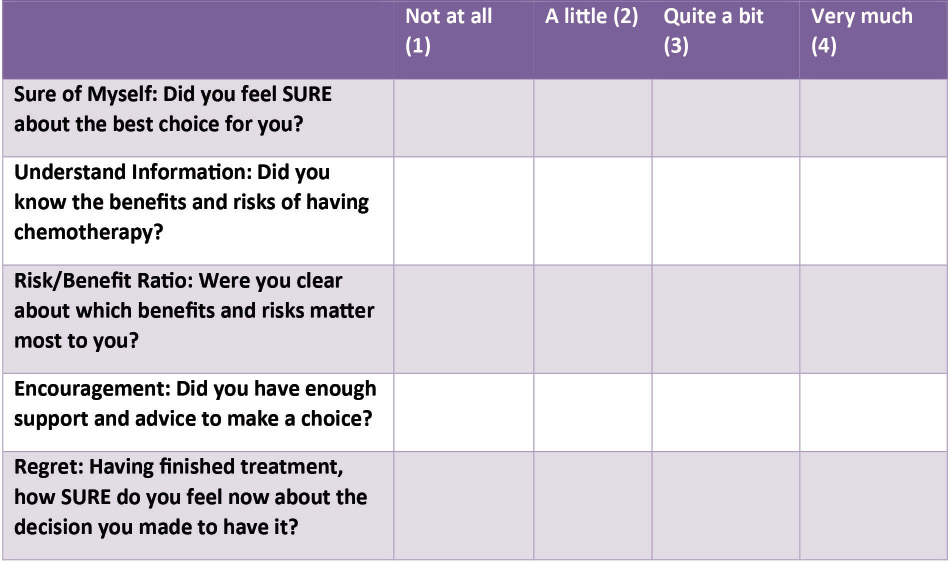
We based our questionnaire on the SURE test, a validated tool developed to identify decisional conflict at the time of decision-making (Figure 1).9 The first four questions were adapted for retrospective use and related to how patients felt at the time about the decision-making process. A fifth question was added, in order to explore how people felt having completed chemotherapy about the original decision. The five questions each had four possible responses, producing a maximum 20-point score indicating strong decisional certainty, both at the time of the original decision and with hindsight.
Figure 1 Questionnaire, adapted from the SURE test
Patients with lung or gynaecological cancers who had completed at least one course of palliative chemotherapy, which typically included several cycles (doses) of treatment, were identified by their clinical teams during routine visits to the ECC. Interviews took place opportunistically, either before or after consultations. Palliative intent is an overarching term for chemotherapy given when the goal is not cure, but instead is to improve symptoms and/or to extend life. Patients were asked if they would be willing to complete the questionnaire with a medical student, BG, following their clinic appointment.
BG completed the questionnaire with patients in a private clinic area. If patients offered unprompted comments or qualifications, these were documented. Unprompted comments provided insights into experiences and reflections on the decision-making process, adding nuance to the numeric SURE test results. We did not capture this data verbatim or undertake formal analysis. However, key issues, statements or terms were noted.
We received approval for the study from NHS Lothian following review by the Clinical Lead for Quality Improvement in the ECC, and ethical approval from the University of St Andrews Teaching and Research Ethics Committee.
Results
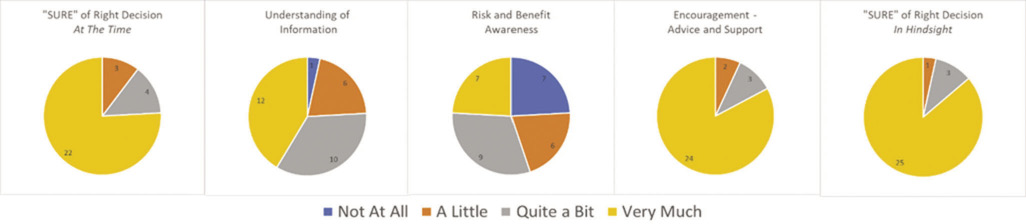
A total of 29 patients completed the questionnaire with BG between during July 2017. Of these, 21 patients had lung cancer, of whom eight were female and 13 male. The remaining eight participants had ovarian cancer. A summary of questionnaire results are presented in Figure 2.
Figure 2 Questionnaire responses in five domains from 29 patients
The decision to undergo chemotherapy
A total of 22 out of 29 (76%) patients reported having felt ‘very sure’ at the time of the decision that chemotherapy was the best choice for them, with the remaining four out of 29 (14%) feeling ‘quite sure’ and three out of 29 (10%) feeling only ‘a little sure’. None reported having felt ‘not at all sure’. All eight patients with ovarian cancer reported having felt ‘very much sure’ that the decision was right for them.
Having undergone chemotherapy, and with the benefit of hindsight, 25 out of 29 (86%) patients remained ‘very much sure’ that their decision to have treatment had been the right one, three out of 29 (10%) were ‘quite sure’ about this and only one patient was ‘not at all sure’. However, unprompted comments from four patients revealed that the decision to undergo chemotherapy had not necessarily felt like a choice.
Didn’t see chemotherapy as a choice, rather a necessary evil
Did not view chemotherapy as a choice, rather felt it was the only option
Chemotherapy was not a choice, but something that had to be done
There was no real choice – not forced but channelled in to it
Clinician support and advice around the decision
A total of 24 out of 29 (83%) patients felt ‘very sure’ that they had received enough support and advice to make the decision. Three out of 29 (10%) reported feeling ‘quite sure’ about this and two patients remembered feeling only ‘a little sure’. None reported having felt ‘not at all’ sure. Unprompted comments reveal that some patients felt that they had not received the type of information that they needed.
Felt there could have been more emphasis on the practicalities of chemo
Would like to have been more informed about the mechanisms by which drugs work
Information was provided in multiple formats but too generalised to be helpful
Wasn’t fully aware of all side effects until second round of chemotherapy
Patient understanding risks and benefits of treatment
There was more heterogeneity in the responses relating to patient understanding of the risks and benefits of chemotherapy. Twelve out of 29 (41%) were ‘very sure’ that they knew the benefits and risks of having chemotherapy, 10 out of 29 (35%) were ‘quite sure’, six out of 29 (21%) ‘a little sure’ and one patient was ‘not at all sure’. This patient had lung cancer and had expressed a low level of confidence in the decision to have chemotherapy at the time.
Fewer patients still reported having understood the risks and benefits of treatment as they applied to their own situation. Only seven out of 29 (24%) patients were ‘very sure’ about which benefits and risks mattered most to them, nine out of 29 (31%) were ‘quite sure’, six out of 29 (21%) ‘a little sure’ and seven out of 29 (24%) were ‘not at all sure’.
One patient seemed not to have grasped that chemotherapy was not being given with curative intent.
Didn’t feel the risks were relevant, because without chemo the risk of dying was so great
A different patient shared their optimism that the chemotherapy would provide meaningful benefit.
If it’s going to help you in the long run, do it
Discussion
The majority of people we surveyed felt confident that the decision to undergo chemotherapy had been the right one, both at the time that they made the original decision and with hindsight. The vast majority also felt well supported and advised by their clinicians in relation to the decision. However, the patient sample included only: patients who had elected to receive chemotherapy; those who had completed their treatment and remained well enough to attend clinic; and, those considered by the clinician to be robust enough to invite to interview. Our relatively positive findings may be due to this selection bias, or could reflect genuine comfort in decision-making in Scottish oncology clinics.
Patients reported less confidence in their understanding of specific risks and benefits of treatment, and less confidence still in how these risks and benefits applied to their own situation. What we do not know is the extent to which patients were offered the opportunity to understand more about risks and benefits, or to consider how these related to them as individuals. It is possible that patients chose to make their decision without this knowledge, but it is also possible that they did not; and that they would have made a different decision had they understood more.
Unprompted comments from patients revealed specific areas where patients’ information and support needs were perhaps less well met. One important area raised by patients’ comments was the extent to which chemotherapy was perceived as a choice. Four patients described having felt that there had been no choice. However, exploring the meaning behind their comments was beyond the scope of our study.
Clinicians cannot assume what a patient’s preference around cancer treatment might be and it is accepted that an exploration of preferences should be part of the oncology consultation.10 Shared decision-making, where people are empowered to be active partners in treatment choices, is a cornerstone of the Scottish Chief Medical Officer’s report, Realistic Medicine.6 In practice, patients attending oncology clinics to discuss the option of palliative chemotherapy may do so with an already established preference to undergo chemotherapy.11 Preconsultation preferences have been shown to strongly predict the likelihood of whether the person with cancer goes on to receive chemotherapy.12 Patients are influenced by a range of people, including those close to them and their professionals, when making a decision about cancer treatment.13 In one study of women with advanced breast cancer, the oncologist was often the most influential person in their decision; and in the majority of cases the oncologist had recommended treatment.14 The reality is that many patients defer to their clinicians when making decisions about treatment and this has implications for the shared decision-making and informed consent process.
We do not know the specifics of what had been discussed in clinic with patients in our study, since we saw them after they had completed their treatment. One study in which oncology consultations around chemotherapy for advanced cancer were recorded revealed that oncologists commonly asked about patient preferences, and often early in the discussion.11 However, this was often at the expense of exploring the values or knowledge that underpinned the expressed preference. A deliberative approach to consultation, where values and information are shared in a model that enables joint preference construction, was proposed; this is in line with the landmark Montgomery ruling relating to informed consent.15
A large North American survey of people with advanced cancer undergoing palliative chemotherapy revealed that a majority (69%) did not understand that their treatment would not offer them cure.16 The authors suggested that oncologists may be able to improve their patients’ understanding of realistic goals of treatment, but also cautioned that this could reduce patients’ satisfaction with their oncologist. This assertion is alarming, and may reflect a reality in the North American private healthcare system where patient satisfaction (even if based on false hope) may be viewed as preferable to honesty about uncomfortable truths.
Another North American study interviewed people who had previously undergone palliative chemotherapy for advanced cancer.17 Patients varied in their reasons for taking chemotherapy, but also in the thresholds for potential survival benefit that were acceptable to them. Worryingly, some patients who had received chemotherapy would have made a different decision had they better understood the facts.
The concept of weighing up risks and benefit is very familiar to medical professionals, but it is something that patients and those close to them can grapple with. In our study, BG often needed to clarify what these terms meant. It is well recognised that patients commonly struggle with statistical information.13 One patient in our study expressed that they did not see risks as relevant to them as they felt that without chemotherapy their risk of dying was so great. In the palliative chemotherapy setting, the reality is that the risks of treatment typically equate to harm as a result of treatment, but as can be seen from this patient’s comment, ‘risk’ is open to misunderstanding. Perhaps the specific wording of the SURE test regarding this area was too vague for the context; however, we expect that the added explanations offered by the researcher addressed this somewhat. The SURE test had previously been validated for use as a ‘real-time’ measure of decisional conflict. Our decision to adapt the questions for retrospective use may threaten the tool’s validity. In addition, we added a fifth question and summed the scores from all questions together.
The lack of a robust real-world evidence relating to both the burden of cancer treatment but also the likely outcomes18 may limit the capacity for fully informed risk/benefit discussions. The American Society of Clinical Oncology and the European Society for Medical Oncology both draw attention to the potential burdens of cancer treatment to individuals and societies that extend beyond physical treatment toxicities; and provide practical guidance on how the value of cancer treatments might be assessed.19,20
A few patients in our study described that the decision to undergo chemotherapy had not felt like a choice. It might be that people knew that cancer treatment was right for them, with the sense of ‘nonchoice’ reflecting that. However, it could be that they were not aware of the alternative, or that the alternative did not appear favourable.
There are several potential factors at play, any of which may contribute to the feeling that cancer treatment is the only option. We have strong societal narratives around battling/fighting cancer,21 promoting the idea that ‘something must be done’. In this context, the option of not receiving cancer treatment may sit uncomfortably. However, the way in which the alternative to cancer treatment is understood and presented must be considered. In the UK and many other countries, ‘best supportive care’ is the term used to describe the plan when people are too unwell for, or choose not to have, cancer treatment. Whilst this sounds like a management plan that might offer something meaningful and helpful, our experience is that this is often not the case, for the most part because there is no agreement about what it means in practice.22 We suggest that unless we can describe what the best supportive care looks like in practical terms, the decision whether to undergo palliative cancer treatment will continue to feel like a nonchoice for many.
A culture within healthcare, where patients who choose not to undergo cancer treatment might be described as ‘refusing’ treatment, is another factor to consider. A decision not to undergo cancer treatment has to be a legitimate option if the informed consent process is to be valid. Tessa Richards, an editor at the BMJ, highlighted the inadequacy of discussions about her own cancer treatment options.23 She describes her experience of living with incurable cancer, and her frustration that the option of not having further cancer treatment was not presented. The time and resource that goes into discussing treatment options for people at early stages of cancer is great, and arguably discussions around palliative cancer treatment and supportive care should be similarly available.24
Oncology consultations can be greatly pressured: taking a full clinical and social history; physical examination; explanation of the diagnosis/prognosis; discussion about risks and benefits of potential treatment/s; and, informed consent if cancer treatment is the plan. Oncologists must also respond and adapt to the emotional reactions of patient and anyone accompanying them. Thus, we appreciate that the time for detailed discussions about risks and benefits of treatment, as well as how these apply to the individual, is very limited.
It is recognised that patients may not describe regret in relation to chemotherapy until several months following its completion.25 Thus, for many patients in this small study, it may have been too early for them to experience this feeling or to report it.
Although we tried our best to ensure anonymity of patients’ responses, we are mindful that some patients may have exhibited response bias in terms of giving socially desirable answers, or leant towards acquiescence.
In conclusion, participants reported high levels of confidence in their decision to undergo chemotherapy, despite simultaneously reporting lower levels of understanding of the risks and benefits and how these were relevant to them. Perhaps patients having the opportunity to receive the information they want is more important than how much information they have chosen/been able to absorb. This approach is more patient-centred than a ‘one-size’ model in which everyone is expected to understand the same level of information.
Further research with larger scale and robust studies would enable a deeper understanding of some of the more complex and nuanced areas highlighted by our findings. These include the factors that contribute to patients viewing the chemotherapy decision positively; the root causes and effects of the contrast between how much people understand and how confident they are in their decision; and what leads some people to choose not to undergo palliative chemotherapy.
Decision-making around palliative chemotherapy for incurable cancer will always pose challenges. The onus is on clinicians to understand the information needs and preferences of the people they care for, and to strive to meet these needs in a personalised way.
Acknowledgements
The authors would like to thank all of the patients who participated in this study. Their generosity in giving up their time and their honesty about their experiences was deeply appreciated. We would also like to thank the clinicians who took the time to identify and approach patients to participate.
References
1 Wallington M, Saxon EB, Bomb M et al. 30-day mortality after systemic anticancer treatment for breast and lung cancer in England: a population-based, observational study. Lancet Oncol 2016; 17: 1203–16.
2 Santoso JT, Engle DB, Schaffer L et al. Cancer diagnosis and treatment: communication accuracy between patients and their physicians. Cancer J 2006; 12: 73–6.
3 Mack JW, Walling A, Dy S et al. Patient beliefs that chemotherapy may be curative and care received at the end of life among patients with metastatic lung and colorectal cancer. Cancer 2015; 121: 1891–7.
4 Prigerson HG, Bao Y, Shah MA et al. Chemotherapy use, performance status, and quality of life at the end of life. JAMA Oncol 2015; 1: 778–84.
5 Kasman DL. When is medical treatment futile? A guide for students, residents, and physicians. J Gen Intern Med 2004; 19: 1053–6.
6 Chief Medical Officer’s Annual Report 2014–15. Realistic Medicine. http://www.gov.scot/Publications/2016/01/3745 (accessed 01/08/18).
7 Leng G, Clark CI, Brian K et al. National commitment to shared decision making. BMJ 2017; 359: j4746.
8 LeBlanc A, Kenny DA, O’Connor AM et al. Decisional conflict in patients and their physicians: a dyadic approach to shared decision making. Med Decis Mak 2009; 29: 61–8.
9 Légaré F, Kearing S, Clay K et al. Are you SURE? Assessing patient decisional conflict with a 4-item screening test. Can Fam Physician 2010; 56: e308–14.
10 Blinman P, Alam M, Duric V et al. Patients’ preferences for chemotherapy in non-small cell lung cancer: a systematic review. Lung Cancer 2010; 69: 141–7.
11 Henselmans I, Van Laarhoven HWM, Van der Vloodt et al. Shared decision making about palliative chemotherapy: a qualitative observation of talk about patients’ preferences. Palliat Med 2017; 31: 625–33.
12 Koedoot CG, de Haan RJ, Stiggelbout AM et al. Palliative chemotherapy or best supportive care? A prospective study exploring patients’ treatment preference and choice. Br J Cancer 2003; 89: 2219–26.
13 Thornton M, Parry M, Gill P et al. Hard choices: a qualitative study of influences on the treatment decisions made by advanced lung cancer patients. Int J Palliat Nurs 2011; 17: 68–74.
14 Grunfeld EA, Maher J, Browne S et al. Advanced breast cancer patients’ perceptions of decision making for palliative chemotherapy. JCO 2006; 24: 1090–8.
15 Chan SW, Tulloch E, Cooper ES et al. Montgomery and informed consent: where are we now? BMJ 2017; 35: j2224.
16 Weeks JC, Catalano PJ, Cronin A et al. Patients’ expectations about effects of chemotherapy for advanced cancer. N Engl J Med 2012; 367: 1616–25.
17 Silvestri G, Pritchard R, Welch HG. Preferences for chemotherapy in patients with advanced non-small cell lung cancer: descriptive study based on scripted interviews. BMJ 1998; 317: 771–5.
18 Booth CM, Tannock IF. Evaluation of treatment benefit: randomized controlled trials and population-based observational research. JCO 2013; 3298–9.
19 Schnipper LE, Davidson NE, Wollins DS et al. American Society of Clinical Oncology statement: a conceptual framework to assess the value of cancer treatment options. J Clin Oncol 2015; 33: 2563–77.
20 Cherny NI, Sullivan R, Dafni U et al. A standardised, generic, validated approach to stratify the magnitude of clinical benefit that can be anticipated from anti-cancer therapies: the European Society for Medical Oncology Magnitude of Clinical Benefit Scale (ESMO-MCBS). Ann Oncol 2015; 26: 1547–73.
21 Granger K. Having cancer is not a fight or a battle. The Guardian. 25 April 2014.
22 Bowden J, Fenning S, Marron B. Best supportive care in advanced lung cancer – more than just a label? BMJ 2018; 363: k5017.
23 Richards T. The responses to the “cancer drugs scandal” must fully involve patients—an essay by Tessa Richards. BMJ 2017; 359: j495.
24 James N. Deciding when to say no more. Cancer Nurs Pract 2014; 13: 12.
25 Hacking B, Wallace L, Scott S et al. Testing the feasibility, acceptability and effectiveness of a ‘decision navigation’ intervention for early stage prostate cancer patients in Scotland – a randomised controlled trial. Psycho-Oncology 2012; 22: 1017–24.
