A 26-year-old male patient from Faridpur, Bangladesh, presented to the Emergency Department with a history of 6 days’ high intermittent fever, 4 days’ fluctuating consciousness and a generalised tonic–clonic seizure (GTCS) a few hours before presentation. Immediately after arrival he had two further episodes of GTCS. After appropriate measures, he was moved to the Intensive Care Unit (ICU) for observation and further management as he had a low Glasgow Coma Scale (GCS) score of E1V2M2, assumed to be postconvulsive state. After about 2 hours, his GCS improved to E3V4M6. However, around 4 hours later, his GCS deteriorated and he had to be intubated for airway protection. Detailed history taking revealed that he drank raw date palm sap (a local delicacy) about 3 days prior to initial symptoms. Cerebrospinal fluid (CSF) study revealed leucocytosis with predominant lymphocytes and raised protein. MRI of brain (without contrast) revealed multiple small hyperintense foci involving the corpus callosum, bilateral cerebral and cerebellar hemispheres, most prominent in fluid-attenuated inversion recovery (Figure 1) and diffusion-weighted imaging (Figure 2). No perilesional oedema or mass effect was evident on the MRI. History, clinical features and laboratory parameters indicated the possibility of encephalitis due to Nipah virus (NiV) infection, which was confirmed by real-time polymerase chain reaction (RT-PCR) from CSF. With supportive management at the ICU, he gradually improved over the next 3 weeks and was subsequently discharged from hospital. At follow up 3 and 6 months later, he was well and without any complication.
Figure 1 MRI without contrast (fluid-attenuated inversion recovery imaging) showing multiple small hyperintense foci involving subcortical and deep white matter of brain
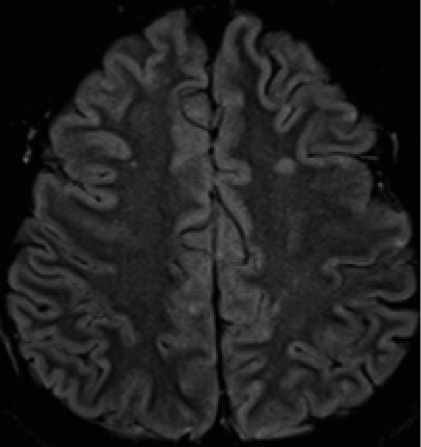
Figure 2 MRI without contrast (diffusion-weighted imaging) showing multiple small hyperintense foci involving subcortical and deep white matter of brain
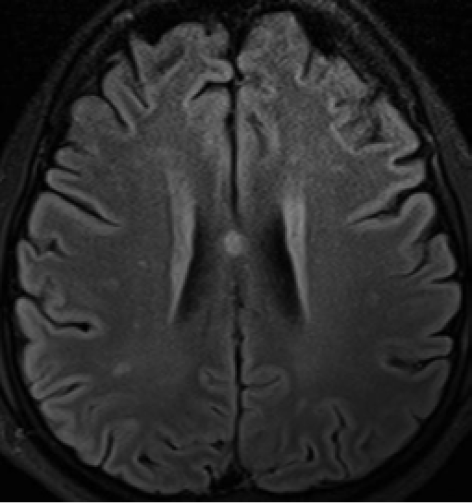
NiV, a zoonotic paramyxovirus, affects the endothelium of medium and small-sized blood vessels, resulting in diffuse vasculitis and vasculitis-induced thrombosis in different vital organs, including respiratory tract, heart and kidneys.1 However, extensive thrombosis and parenchymal necrosis are mostly significant in the central nervous system, as evident from histopathological findings from autopsy of patients with fatal NiV infection.2 The multiple small scattered hyperintense lesions on the MRI, especially in the white matter of brain, indicate microvascular infarction, consistent with the pathophysiology of NiV infection. Thus, NiV encephalitis has distinctive MRI findings, especially in the acute stage, unique from other encephalitides.3,4 However, a different MRI pattern (multiple confluent patchy cortical involvement) is seen in patients with delayed onset and relapse of NiV, probably due to the extensive and irreversible damage from the vasculitis.1,3,4
NiV has been recognised to cause outbreaks of fatal encephalitis in Bangladesh and neighbouring India, almost every year since 2001.5 Ever since its identification during the outbreak in Malaysia and Singapore in 1998–99, NiV has been attributed as a health hazard owing to its easy transmissibility from animals (bats and pigs), contaminated food (by animal saliva or excreta, e.g. raw date palm juice) or directly from human-to-human, and its high fatality rate (40–75%). Because of the distribution of its natural host, fruit bats (of Pteropodidae family) and other similar bats, several south east Asian and east African countries are at potential risk for infection.1,5 Incubation period usually ranges from 4–14 days, but has been reported up to 45 days.5 Clinical presentation in humans ranges from asymptomatic (subclinical) infection, to coryzal features, high fever, respiratory manifestations (mild to severe) and altered level of consciousness. Seizures and fatal encephalitis occur at extreme severity.1,5 The confirmatory diagnostics available are RT-PCR from body fluids and antibody detection via enzyme-linked immunosorbent assay.5 Unfortunately, no drug or vaccine specific for NiV is available to date, which makes prevention the only means to fight against this disease. Avoiding consumption of fresh/raw date palm juice, using personal protective gear while handling infected individuals, handling of clinical samples by trained personnel in properly equipped facilities, etc. are some of the most effective tools for preventing NiV infection.1,5
With the long incubation period, there is possibility of NiV spreading in different regions through travellers, especially in this era of airplane travel. Some authors have also expressed their worries about the use of this agent for bioterrorism.1,3 Only well-informed and vigilant clinicians can counter the potential threat from this deadly organism. Unlike MRI, availability of the confirmatory diagnostics are very limited. During an infection outbreak, given the appropriate epidemiological and clinical settings, MRI can be invaluable for the early detection of the disease and to differentiate from other causes of encephalitides.
References
1 Ang BSP, Lim TCC, Wang L. Nipah virus infection. J Clin Microbiol 2018; 56: e01875-17.
2 Wong KT, Shieh WJ, Kumar S et al. Nipah virus infection: pathology and pathogenesis of an emerging paramyxoviral zoonosis. Am J Pathol 2002; 161: 2153–67.
3 Lim TCC, MR imaging in Nipah virus infection. Neurol Asia 2009; 14: 49–52.
4 Sarji SA, Abdullah BJ, Goh KJ et al. MR imaging features of Nipah encephalitis. Am J Roentgenol 2000; 175: 437–42.
5 World Health Organization. WHO fact sheet: Nipah Virus. 2018. https://www.who.int/news-room/fact-sheets/detail/nipah-virus (accessed 17/03/19).
