A female in her forties with glaucoma was admitted with a cluster of focal epileptic seizures consisting of impaired awareness, forced head turning to the left, and jerking of the left upper and left lower limbs. Following stabilisation, neurological examination detected a left homonymous hemianopia and mild left hemiparesis. The focal nature of her seizure semiology and persisting neurological signs suggested the possibility of a right hemispheric structural lesion, thus further investigation with neuroimaging was required.
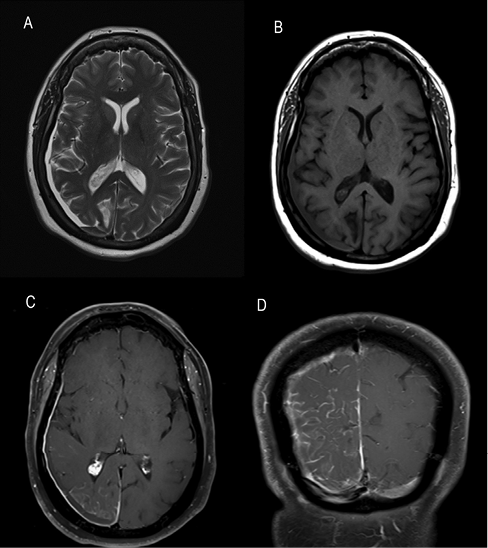
CT of her brain revealed asymmetric mild atrophy affecting the right occipital lobe (Figure 1a) and right parietal lobe (Figure 1b), and foci of right gyral calcification (Figure 1b). An incidental small right subdural haematoma was also apparent on MRI of her brain (Figure 2a and 2b). More importantly, gadolinium contrast administration showed abnormal enhancement of the right intracranial meninges, both outer (dural) and inner (leptomeningeal) leading to a striking serpentine pattern of enhancement filling the right parieto-occipital sulci (Figure 2c and 2d). This characteristic constellation of imaging abnormalities allowed us to reach a diagnosis of Sturge–Weber syndrome (SWS), despite careful clinical inspection of her skin showing no abnormality.
Figure 1 (a, b) CT brain axial images
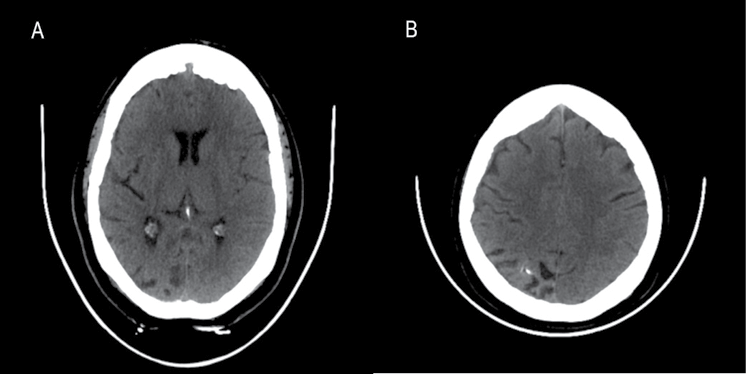
Figure 2 MRI brain axial (a) T2 and (b) T1 images, and T1 contrast-enhanced (c) axial and (d) coronal brain images
SWS is a sporadic neurocutaneous disorder classically encompassing the recognised triad of facial port-wine nevus, ipsilateral intracranial involvement (leptomeningeal angiomatosis) and glaucoma.1 Diagnosis is generally straightforward since the port-wine nevus is both characteristic and difficult to overlook. Facial nevus can occur without intracranial abnormalities (type 2 SWS) and rarely, in around 10%, leptomeningeal angiomatosis manifests without the distinctive port-wine nevus (type 3 SWS).1,2 Clinical identification of type 3 SWS is more challenging, as in the absence of cutaneous involvement, accurate diagnosis relies primarily upon an awareness of type 3 SWS and recognising compatible imaging evidence of leptomeningeal angiomatosis.
Typical neuroimaging features of SWS as exemplified by our patient include cerebral hemiatrophy (most often affecting parietal and occipital lobes), ipsilateral gyral calcification, ipsilateral prominent choroid plexus (also seen in Figure 2c) and ipsilateral leptomeningeal angiomatosis visualised as vascular serpentine contrast enhancement.3 Neurological features may include seizures, headaches, stroke-like episodes, hemiparesis, visual field defects and cognitive impairment. These most often manifest in childhood, although adult presentations are also recognised.4 Glaucoma and endocrine abnormalities, particularly growth hormone deficiency and central hypothyroidism, are also encountered. Multidisciplinary management involving neurology, ophthalmology, dermatology and endocrinology input is essential.4
Our case demonstrates that a high index of suspicion allows recognition of SWS without any cutaneous involvement, in the appropriate clinical context and supported by characteristic neuroimaging features described above.
Acknowledgement
The authors would like to thank Professor Mohamed Jan whose Neuroscience Forum enabled collaboration on this case.
References
1 Sudarsanam A, Ardern-Holmes SL. Sturge-Weber syndrome: from the past to the present. Eur J Paediatr Neurol 2014; 18: 257–66.
2 Comi AM. Sturge-Weber syndrome. Handb Clin Neurol 2015; 132: 157–68.
3 Nandigam K, Mechtler LL, Smirniotopoulos JG. Neuroimaging of neurocutaneous diseases. Neurol Clin 2014; 32: 159–92.
4 Bachur CD, Comi AM. Sturge-Weber syndrome. Curr Treat Options Neurol 2013; 15: 607–17.